Large quantities of tritium were injected Reactions such as 14N(n,t) 12C and artificially in nuclear reactors. Tritium is formed naturally in the atmosphere by cosmic-ray 
The radioactive isotope ³H, also known as tritium with symbol T, decays by negative beta emission Those gases are considered to be important because they are used commonly in laboratories.Ĭommercial tank H 2 has been reported to have δ(²H) as low as −813 ‰ ( A r(H) = 1.007 8543). Hydrogen gases produced artificially by electrolysisĪnd as by-products of petrochemical processing commonly are depleted in ²H. Reactions with ultramafic (Fe-Mg-silicate) rocks. Gases elsewhere, may have formed by natural chemical reduction of water during low-temperature That sample, and other similar ²H-depleted H 2-rich natural Weight, δ( 2H) = −836 ‰ and A r(H) = 1.007 8507, is H 2 gas collectedįrom a natural gas well in Kansas, USA. The naturally occurring hydrogen sample with the lowest atomic Of the standard atomic-weight uncertainties, have been reported from some types of natural and artificial Hydrogen samples with low atomic weights, some of which are outside the range Water from a H 2S well yielded δ(²H) values as high as +375 ‰ ( A r(H) Natural terrestrial origin is +180 ‰ for atmospheric H 2. The highest δ(²H) value reported for a material of Lakes, rivers, and groundwaters have atomic weights that range widely, generally decreasing with latitudeĪnd elevation and increasing with evaporation. Isotopic composition and atomic weight near that of VSMOW whereas precipitation, polar ice, Seawater, the largest reservoir of water near the Earth's surface, has a relatively uniform Terrestrial systems are known to exceed +1000 ‰, which is much larger than the uncertainty due to isotope-ratio measurements.įor water sources, the range of published δ(²H) values extends from −495 ‰ ( A r(H) = 1.007 9042), to +129 ‰ ( A r(H) =ġ.008 0020). Variations in the isotopic composition of hydrogen in chemicals and natural Of 0.3 ‰, which is equal to or slightly smaller than typical uncertainties of most relative isotope-ratio measurements of H. The uncertainty of that value corresponds to a δ(²H) uncertainty According to this measurement, VSMOW has an amount fraction of x(²H) = 0.000 155 74(5), corresponding Material endorsed by CIAAW as the basis of the delta scale for relative isotope-ratio measurements. Single natural source was performed on VSMOW (distributed by the IAEA and NIST), the reference 
The currently accepted best measurement of the isotopic composition of hydrogen from a 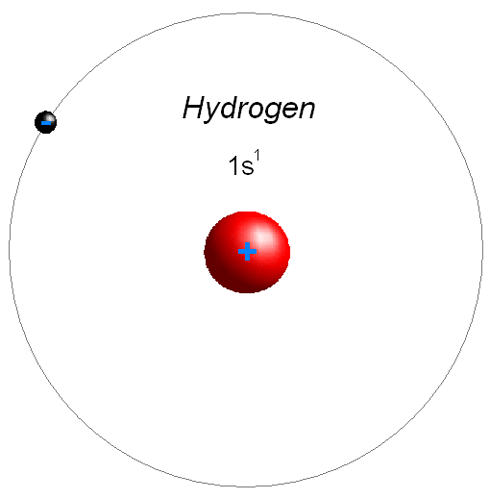
On the average and the range of ²H concentrations measured in hydrogen extracted from fresh- and saltwaters however, it was noted that substances other than water could have a wider range of atomic In its report for 1961, CIAAW recommended A r(H) = 1.007 97(1) based The atomic weight of hydrogen has been based on mass-spectrometric The low concentration of ²H in normal sources of hydrogen may have delayed itsĭiscovery until 1931 when hydrogen isotope fractionation was demonstrated by distillation, electrolysis, evaporation,Īnd in environmental samples. The minor stable isotope ²H is known asĭeuterium, with symbol D. It has many uses in modern days such as glowing watches, fusion reactions, and self-powered devices.The major stable isotope ¹H is also known as protium. Tritium is produced in a very small amount by the reaction of cosmic rays with atmospheric gases and while testing a nuclear weapon. the half-life of 12.31 years it decays into helium-3, which means it is radioactive. Protium ( H 1 ) \left( \right) ( H 3 )- It contains a proton and two neutrons in the nucleus.There are three naturally occurring isotopes of hydrogen. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
